Group Research in the News: Song secures $2.89-million grant to study drug repositioning for Alzheimer’s disease; $2.1 million grant seeks to identify potential therapeutic strategy for Alzheimer’s disease
Research Statement
Our research goal is to identify therapeutic drug candidates for target proteins involved in Alzheimer’s disease and other diseases using integrated unbiased virtual screening/translational bioinformatics approach, biophysical and biological experimental validation. We also investigate protein-protein interactions, small molecule-target protein interactions and its mediated cellular signaling events, and how these are changed by disease-associated genetic variants to gain insights for the mechanisms of action in Alzheimer’s disease and other diseases. Methodology development for multiscale modelling of complex biological systems continues to be a focus of my research group. In addition to $2.1 million NIH R01 awarded to study TREM2-endogneous ligand interactions in Alzheimer’s disease through 2021 – 2026; we are recently awarded $2.89 million NIH R01 grant to study drugs repositioning for Alzheimer’s disease through 2023 – 2028.
Research projects in my group
1. Identify endogenous and exogenous small molecules, including reposition of FDA-approved drugs, for TREM2 protein and determine its functional mechanisms in Alzheimer’s disease, offering a molecular basis for strategy development to better target TREM2 for Alzheimer’s disease treatment (Dean et al, 2019 Frontiers in Neurology; Kober et al, 2020 Alzheimer’s and Dementia)

Fig. Neurodegenerative Disease–Associated Variants in TREM2 Destabilize the Apical Ligand-Binding Region of the Immunoglobulin Domain
Single amino acid variants in triggering receptor expressed on myeloid cells 2 (TREM2) have been identified by genome-wide association studies to be one of the strongest genetic risk factors for late-onset Alzheimer’s disease (AD). Neurodegenerative disease–associated variants including AD-associated variants in TREM2 impair TREM2’s ability to bind and signal in response to endogenous and exogenous ligands. The molecular mechanisms of TREM2 in AD remain not fully understood, in part due to the lack of conclusive identities for TREM2’s ligands and the lack of a good understanding of how different ligands interact with TREM2 to affect TREM2 functions. The research goal is to identify endogenous and exogenous small molecules, including reposition of FDA-approved drugs, for TREM2 protein and determine its functional mechanisms in Alzheimer’s disease using integrated computational modeling, biophysical and biological experiments. Results gained from this study will further the insight into TREM2 immune activation by endogenous and exogenous ligand binding, offering a molecular basis for strategy development to better target TREM2 for AD treatment.
Collaborators: Drs. Erik Roberson (UAB), Tom J. Brett (WUSTL) and Li Gan (Weil Cornell Medicine)
Funding Support: National Institutes of Health (NIH) and Alzheimer’s Drug Discovery Foundation
2. Protein interactions underlying death receptor-mediated apoptosis and identification of molecular targets to overcome the drug resistance in breast cancer (Suever et al, 2008; Pan et al, 2011; Yan et al, 2013, Fancy et al, 2014, Yang et al, 2016, Fancy et al, 2016, 2017, 2018)
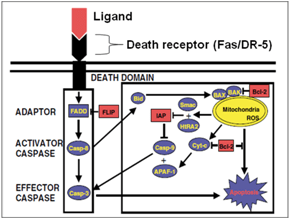
Breast cancer is the second leading cause of cancer death in the United States. The disease affects an estimated 1.3 million women annually worldwide. Chemotherapy treatments for breast cancer have steadily increased the survival rate, however, drug resistance, toxicity and poor drug response are critical issues that still hinder the effective treatment of breast cancer. Death receptor-induced signaling complex (DISC) has been shown to regulate apoptosis in a variety of cell types, including breast cancer. DISC formation is a critical step in death receptor-mediated apoptotic signaling (Figure 1). The goal of this project is to determine new protein target that could regulate the death-receptor mediated apoptosis and indentify key molecular markers for the cross-talk between apoptosis and cell survival pathways in breast cancer. The results of this study could unveil the potential target(s) to regulate death-receptor-mediated apoptosis and aid in the development of strategies to increase chemotherapeutic drug efficacy and overcome the drug resistance in breast cancer treatments.
Collaborators: Drs. Donald J. Buchsbaum, Harrison Kim, Kurt R. Zinn, Catherine C. Parker, Tong Zhou, John Mountz, Jay M McDonald
Funding Support: NIH
3. Molecular regulatory mechanisms for calreticulin-modulated signaling in focal adhesion disassembly, collagen expression and apoptotic cell removal, and for glycosylated integrin-mediated signaling in cell adhesion for tissue remodeling
Wound healing in response to injury requires well-coordinated integration of the complex biological and molecular events associated with cell adhesion, cell migration, apoptotic removal and extracellular matrix remodeling. Regulation of these cellular processes represents points of intervention for therapeutics for deficient wound healing or excessive repair resulting in fibrosis and scarring.
Thrombospondin and calreticulin-modulated signaling in focal adhesion disassembly (Yan et al, 2010, 2011, Wang et al, 2014, 2018, 2019, 2020, Yang et al 2020).

Fig. Thrombospondin and calreticulin mediated signaling pathway for focal adhesion disassembly (Rege et al, 2006)
Binding of the matricellular protein thrombospondin-1 (TSP1) to the cell surface protein calreticulin (CRT) promotes association of CRT with the LDL receptor-related protein (LRP1) to stimulate focal adhesion disassembly (Fig. 2). Loss of focal adhesions can then lead to the intermediate adhesive phenotype, cell migration, and resistance to anoikis (Goicoechea et al, 2003, Pallero et al, 2008). TSP1 signaling through the CRT-LRP1 complex is important for cellular responses to injury and for tissue remodeling through stimulation of intermediate adhesion, survival, and cell motility. However, the molecular mechanisms and structural basis underlying the role of the TSP1/CRT interaction in CRT-mediated focal adhesion disassembly remain unclear. The goal of these studies is to use integrated computational biology and experimental approaches to understand the structural basis and molecular mechanisms underlying the regulation of CRT-induced focal adhesion disassembly by the TSP1/CRT interaction. Results from these studies will facilitate the identification of novel strategies and drugs capable of effectively regulating the focal adhesion disassembly signaling pathway for modulation of tissue remodeling and disease treatments.
Collaborators: Drs. Joanne Murphy-Ullrich and Aaron L. Lucius
Funding Support: National Science Foundation (NSF)
Glycosylated integrin-mediated signaling in cell adhesion (Vomund et al, 2008; Liu et al, 2008; Pan et al, 2010, 2011; Wang et al, 2017).
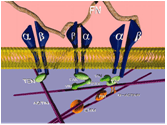

Variations in N-glycosylation of integrins have emerged as an important mechanism for regulation of integrin binding and activity (Gu et al, 2004). Binding of the glycoprotein α5β1 integrin to the extracellular protein fibronectin is critical for multiple cellular functions (Bellis et al, 2004). Altered composition of N-glycans on β1 integrin have been directly linked to changes in cell adhesion, cell spreading, and cell migration. However, the molecular mechanisms underlying the regulation of β1 integrin binding to fibronectin by N-glycosylation of β1 integrin remain unknown. The goal of this study is to characterize the effects of variations in N-glycosylation of β1 integrin on the structural, dynamic, and energetic mechanisms of the β1 integrin-fibronectin interaction using integrated computational biology and experimental approaches. The results from these studies will facilitate the identification of novel strategies to regulate the β1 integrin glycan profile in order to modulate β1 integrin binding to fibronectin. Modulation of this interaction can then be used to further regulate cell adhesion and other cellular activities (Fig. 3), thereby providing novel therapeutic interventions for cardiovascular disease and other disorders.
Collaborators: Drs. Susan L. Bellis (UAB), William Frazier (WUSTL)
4. Novel biomaterial designs for biomedical applications
Peptide self-assembly into collagen-mimetic microfibers (Rele et al, 2007).
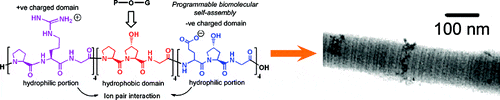
Fig. Peptide self-assembly into D-periodic collagen-mimetic microfibers (Rele et al., 2007)
Collagen is ubiquitously expressed in connective tissue and bone. The triple-helical domain of native collagen is comprised of a strict repeat of the tripeptide sequence Xaa-Yaa-Gly. This sequence leads to a precise organization of individual amino acids in adjacent helices within a defined oligomerization state, imparting structural specificity and fibril-forming properties (Rele et al, 2007). The goal of these studies is to characterize the molecular mechanisms required for peptide assembly into defined, linear assemblies and to determine the optimal peptide design and environment required for the assembly of collagen-mimetic microfibers (Fig. 4). The capacity to mimic collagen’s ability to form linear assemblies with precisely-defined periodic features represents an important milestone in the design of ordered materials for diverse applications in medicine and bionanotechnology.
Cytotoxicity resulting from the alterations of biomembrane mechanics and electrostatics by biomaterials and small molecules (Song, et al, 2005; Wilson et al, 2011).
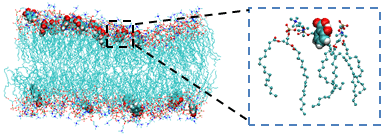
Fig. Interactions of lipid bilayer with small molecule (Song et al, 2005)
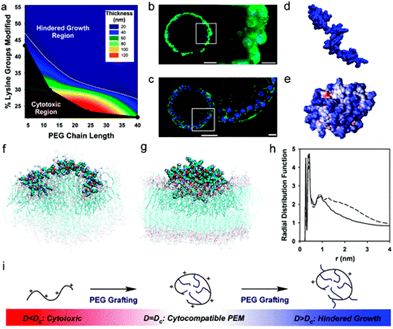
Fig. Cell Surface Engineering with Polyelectrolyte Multilayer Thin Films (Wilson et al, 2011)
Biological membranes play an important role in cellular function by regulating the components of the intra- and extracellular media. Knowledge regarding biomembrane structure, mechanics, and electrostatics is crucial to understanding cellular functions, including permeability, fluidity, deformability, adhesion, and function of membrane-bound proteins. Transplantation has shown great potential for the treatment of many human diseases. Therefore, the design of novel biomaterials for application in immunoisolation nanotechnology to protect transplanted cells from the host immune system provides a promising approach for cell-based therapeutics. The goal of these studies is to understand the changes in biomembrane mechanics and electrostatics that result from interactions between biomembranes and biomaterials or small molecules (Fig. 5, 6). Results from these studies will facilitate the design of optimal biomaterials and small molecules with minimal toxicity for cell-based therapeutics.
Collaborator: Dr. Elliot L. Chaikof (Harvard University)
5. Methodology development for multiscale modeling (Song et al, 2004 a, b; Zhang et al, 2005; Chen et al, 2007)
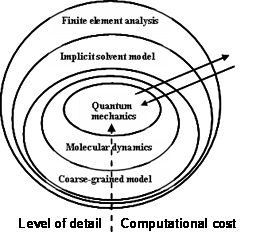
Fig. Multiscale numerical methods across scales
Over the past few decades, different numerical methods at different time and lenght scales (Fig. 7) have been developed and remain under active development. However, several challenges remain for the implementation of multiscale modeling of biological systems across different scales. With respect to these challenges, several questions remain to be answered, including the following:
1) What is the best approach for integration of information across different scales?
2) How should different numerical methods interface?
3) How can mechanics, electrostatics, and heat transfer effects be coupled in multiscale models across scales?
In order to integrate information across different scales and to interface different numerical methods, in addition to using existing software, we will develop new methods for the integration of models, software, and methods across scales. Methods developed in this study will directly contribute to the application studies outlined in other projects.
Copyright ©Yuhua Song, Ph.D.
