Current Research Projects
Our research program is focused on understanding how immune cells and immune mediators impact renal function and end-organ damage. We are especially interested in the interplay between immune cells and the endothelin system. We currently have 3 main lines of investigation in the lab:
ROLE OF ENDOTHELIN IN HYPERTENSION-MEDIATED INFLAMMATION AND END-ORGAN DAMAGE
Individuals that regularly consume high salt diet have a greater risk of developing cardiovascular diseases (CVD) compared to individuals on low salt diet. Moreover, dietary consumption of high salt and the associated inflammation have critical roles in the development of CVD, such as hypertension.
Inflammation, in particular T cell inflammation, is intimately linked to the development of hypertension; however, there is a gap in our knowledge of the mechanisms involved in salt-mediated T cell activation, end-organ damage and CVD risk. Levels of the potent vasoactive peptide endothelin-1 (ET-1) are elevated after high salt consumption and ET-1 has pro-inflammatory actions in the kidney. In this project we aim to elucidate novel molecular pathways involved in the control of the renal pathophysiology associated with increased blood pressure. We will test that 1) activation of the ET-1/ETA axis leads to kidney T cell activation during salt-sensitive hypertension and results in salt-induced kidney damage and CVD risk, and 2) elevated vascular endothelium-derived ET-1 is responsible for the kidney damage observed during high salt consumption.
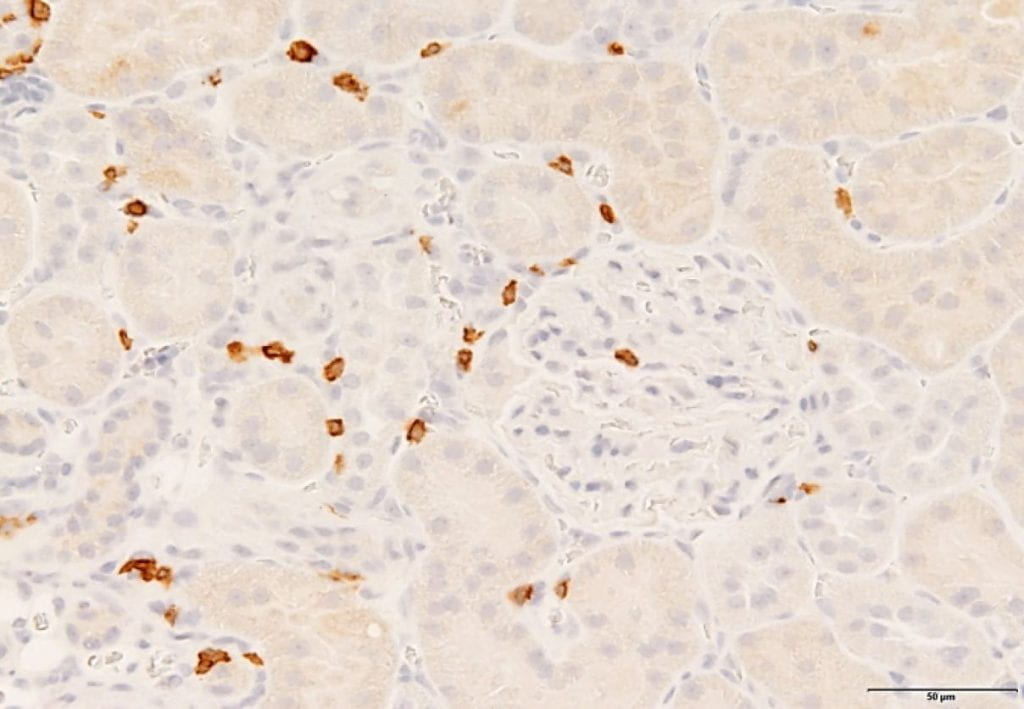
ENDOTHELIN-1 AND RENAL INFLAMMATION IN DIABETIC KIDNEY DISEASE
Diabetes is the leading cause of chronic kidney failure and end-stage renal disease worldwide. One of the hallmarks of diabetic kidney disease is renal inflammation. Endothelin-1 (ET-1) has pro-inflammatory actions in the kidney and induces kidney damage during diabetes via activation of the ETA receptor;
however, the mechanism(s) by which ET-1 mediates these effects remain unknown. Dendritic cells (DCs) are members of the innate immune system and initiators of the inflammatory cascade. Although DCs have been linked to other kidney diseases, their role in the development of kidney damage during the early stages of diabetic kidney disease is unclear. Interestingly, DCs express ET-1 receptors and produce ET-1 upon activation; however, no study has yet determined the role of the ET system in the activation of kidney DCs during diabetes and their role in the development of the associated kidney damage.
The overall hypothesis of this project is that hyperglycemia induces activation of DCs in the kidney in an ETA receptor-dependent manner, leading to diabetic kidney damage.
MECHANISMS INVOLVED IN HYPEROXIA-INDUCED KIDNEY DAMAGE

Oxygen supplementation saves lives, but prolonged exposure to oxygen can also lead to organ toxicity and end-organ damage. This is especially evident in the lungs, but damage to the kidneys has also been described. In this project we are interested in investigating the mechanisms involved in the kidney damage induced by high oxygen exposure, and in particular, the role that endothelin-1 plays in this process.
